Product Highlight
Condition targeted:
Type 2 diabetes

How it works:
d-Nav combines an FDA-cleared mobile app enabled by AI technology, and virtual clinical support to make autonomous adjustments to a patient’s insulin prescription based on their historical and current glucose levels.
Impact:
In a clinical study, 90% of patients saw lower A1C levels in 90 days.*
* Schneider, J. E., Parikh, A., & Stojanovic, I. (2018). Impact of a Novel Insulin Management Service on Non-insulin Pharmaceutical Expenses. Journal of Health Economics and Outcomes Research, 6(1), 53–62. https://doi.org/10.36469/9783
Product Overview
Medical condition:
Type 2 diabetes
Target patient population:
Patients who inject insulin to manage their type 2 diabetes.
What to expect:
Significant improvement in A1c along with a reduction in the frequency of hypoglycemia when used with outpatient therapy.
Note: for more information, please visit https://d-nav.com.
Clinical Overview
Indications for use:
The d-Nav Technology calculates the next dose of insulin to aid in optimizing insulin management. The d-Nav Program is indicated for adults with type 2 diabetes who are injecting insulin.
Outcomes:
In a multi-site randomized control study, d-Nav Technology improved A1C without increasing the risk for hypoglycemia.**
Directions:
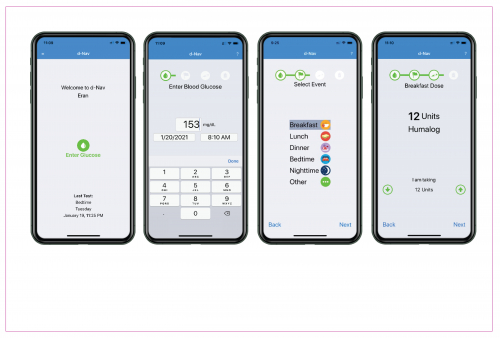
The d-Nav Program consists of FDA-cleared software and unlimited clinical support to optimize insulin dosing. Patients use the d-Nav Technology before every insulin injection by entering their most recent sugar reading, and then receiving a personalized dose recommendation. Data is sent to the cloud where it is monitored by d-Nav Care Specialists, who are available to support patients’ good use and address any clinical conditions.
The use of d-Nav is quick, simple, and easy. Clinical studies show long-lasting A1C reduction sustained for over three years.***
Risks & warnings:
d-Nav is not indicated for patients under 18, or those with gestational diabetes.
Place in therapy:
Complementary to current therapy. Because d-Nav Technology is FDA cleared to autonomously adjust insulin dose recommendations, patients do not need to see their doctor for dose adjustments.
** Bergenstal, R. M., Johnson, M., Passi, R., Bhargava, A., Young, N., Kruger, D. F., Bashan, E., Bisgaier, S. G., Isaman, D. J. M., & Hodish, I. (2019). Automated insulin dosing guidance to optimise insulin management in patients with type 2 diabetes: a multicentre, randomised controlled trial. The Lancet, 393(10176), 1138–1148. https://doi.org/10.1016/s0140-6736(19)30368-x
*** Harper, R., Bashan, E., Bisgaier, S. G., Willis, M., Isaman, D. J., & Hodish, I. (2018a). Temporary Reductions in Insulin Requirements Are Associated with Hypoglycemia in Type 2 Diabetes. Diabetes Technology & Therapeutics, 20(12), 817–824. https://doi.org/10.1089/dia.2018.0266
Product Access
Product description:
Like the closed loop system for patients using a pump, d-Nav Technology seamlessly uses a glucose reading and calculates the next dose of insulin appropriate for the patient. Then, the patient simply injects the recommended dose. Information regarding a patient’s glucose readings and insulin recommendations are always available to the physician via a dedicated provider portal. This program improves outcomes and safety of insulin treatment. d-Nav is safe, precise, and highly effective.
Prescription status:
A prescription from a qualified healthcare provider is required.
Patient access:
Patients download the d-Nav app from the Apple or Google App Store. An enrollment code is generated for patients after their healthcare provider completes a prescription with their personalized treatment details. From that point forward, d-Nav Technology will tell the patient what dose to use before every injection. This takes the guesswork out of managing insulin dosing for the patient. Our trained clinical staff will follow up with the patient regularly to reinforce good use and address clinical concerns.
Use of this product requires access to:
- Internet or wifi – only for initial download and periodic syncing
- A mobile phone
Provider access:
A physician’s office will have the ability to track all the interactions the d-Nav team has with any of their patients through a proprietary, secure physician portal. They will have access to simple progress reports, as well as more detailed reports for each of their patients.
Coverage options:
The d-Nav program is covered by Medicare, Medicaid and most commercial insurance carriers.
Product availability:
d-Nav is available in:
- USA: FDA-cleared Class II Medical Device
- EU: CE mark credentialed by BSI (Class IIa)
- UK: UKCA mark credentialed by BSI (Class IIa)
A better way to use Insulin.
d-Nav Technology is the only autonomous, (level 3) FDA-cleared product that automatically interprets data and autonomously adjusts the treatment plan. d-Nav adjusts an insulin prescription directly to patients, as often as needed.
Clinical Trials
The provided set of evidence represents a sample of conducted studies. For a comprehensive collection contact manufacturers directly.
- Study Title: Automated insulin dosing guidance to optimise insulin management in patients with type 2 diabetes: a multicentre, randomised controlled trial
Study Design: Multicentre, randomized controlled study.
Outcomes: A total of 181 patients were enrolled. The mean decrease in HbA1c from baseline to 6 months was 1.0%±1.0% in the d-Nav group, and 0.3%±0.9% in the control (p<0·0001). The frequency of hypoglycemic events per month was similar between the groups (0.29±0.48 events per month in the intervention group vs 0.29±1.2 in the control group; p=0.96). - Study Title: Temporary Reductions in Insulin Requirements Are Associated with Hypoglycemia in Type 2 Diabetes
Study Design: Observational study
Outcomes: a total of 246 patients using d-Nav were enrolled and followed for 2.8±0.9 years. Significant reduction of total daily insulin was experienced by 70.3% of the patients, occurred 0.8±0.5 times per year, lasted 10.0±7.7 weeks, and insulin requirements declined by 39.9%±12.6%. The frequency of hypoglycemia (<54 mg/dL) was low, at 0.5±0.6 per month, and the difference in frequencies in biphasic/premixed and basal-bolus insulin regimens was not statistically significant. Hypoglycemia was 6.5 times more prevalent during reductions in total daily dose of insulin. - Study Title: Impact of a Novel Insulin Management Service on Non-insulin Pharmaceutical Expenses
Study Design: Prospective cost analysis
Outcomes: Of the 192 d-Nav using patients who completed the 90-day study period, 54 (28.13%) were prescribed one or more expensive medications at baseline, but 45 (83.33%) of those patients were eligible for medication discontinuation after 90 days. Direct savings from medication elimination was estimated to be $145 per patient per month or $1736 per patient per year for all patients and $514/$6172 for the target group. Patients that completed the 90-day period significantly reduced HbA1c levels from 9.37% (CI:7.72%-11.03%) to 7.71% (CI: 6.70%-8.73%)(p<0.001). A total of 170 (88.54%) patients had improved HbA1c at 90 days. - Study Title: Challenging the 50-50 Rule for the Basal-Bolus Insulin Ratio in Patients with Type 2 Diabetes Who Maintain Stable Glycemic Control
Study Design:Observational study
Outcomes: A total of 306 patients with type 2 diabetes who have used d-Nav for at least 6 months for basal-bolus insulin therapy were followed for 3.4 ± 1.8 years. Initial HbA1c was about 9.3% and in less than a year it stabilized at about 7%. In just over three quarters of the cohort, the average basal insulin fraction was <50%; in half of the cohort average basal insulin fraction was <41.2%; and in one‐quarter the basal insulin fraction was <33.6%. Further, the basal insulin fraction did not remain static over time. In half of the patients, the basal insulin fraction varied by ≥1.9×; and, in 25% of the patients, ≥2.5×. - Study Title: Time in range in patients with type 2 diabetes who are long-term users of d–Nav®, an artificial intelligence-driven technology for autonomous titration of insulin dosing
Study Design:Observational study
Outcomes: We acquired CGM information from 18 patients with type 2 diabetes who had been managed on d-Nav® for 2.2 ± 1.0 years. The average glucose level on d-Nav® was 157.1 ± 29.6 mg/dl, and the glucose management indicator or CGM-reported HbA1c was 7.0% ± 0.6. Time in range (TIR, 70-180mg/dl) was 69.8% ± 18.5% of the time. Time below range (<70mg/dl) was 1.8%. Time below 54 mg/dl was 0.3%. The coefficient of variation or glycemic variability was 31.1% ± 6.4%. The sensors were used 76.0% ± 26.6% of the time.
Note: for more research, please visit https://d-nav.com/research/
“d-Nav” refers to the d-Nav Insulin Management Program. “d-Nav Technology” refers to Hygieia’s FDA cleared proprietary mobile app.