Product Highlight
Condition targeted:
Type 1 and 2 Diabetes*, Pre-Diabetes, Hypertension, Heart Failure, Weight & Obesity Management, with integrated mental wellbeing and sleep support

How it works:
The Welldoc Platform includes:
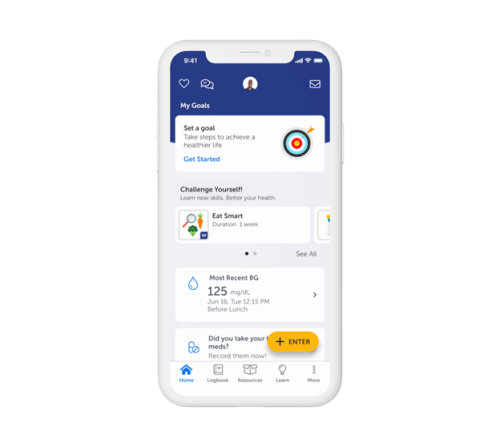
- The Welldoc App – Multi-condition digital coaching across diabetes, hypertension, heart failure, weight management, and prediabetes, with integrated mental wellbeing and sleep support. The app connects and analyzes an individual’s vital health data to provide personalized, AI-driven digital coaching and insights.
- The Welldoc Care Management Portal – Accessible health data and actionable insights to support clinical decisions, interventions, and population health management between office visits.
- Health Reports – Analysis of Welldoc App user-generated data with integrating standards of care to provide insights, trends, and patterns.
Impact:
- 9% Points A1C improvement1
- ≥2% Points A1C reduction with CGM2
- 2-12 mmHg Blood pressure reduction3
- 68% of users showed an average of 6% weight loss over 6 months4
- Based on individuals who paired with weight scales/data over 6 mo. across multiple client programs
- $3,048-$3,252 Cost Savings Per Person with Diabetes5
- 40 minutes saved per patient per year with the Welldoc App6
*Welldoc® Diabetes and Welldoc Diabetes Rx is an FDA-cleared medical device, intended for use by healthcare providers and their adult patients with type 1 or type 2 diabetes. For full labeling information, visit www.welldoc.com. The other Welldoc App products are non-FDA-cleared and intended to promote general wellness and education/self-management of various chronic disease states.
Product Overview
Medical condition:
Welldoc’s comprehensive digital health platform provides cardiometabolic condition digital coaching across pre-diabetes, diabetes, hypertension, heart failure, weight and obesity management, with integrated mental wellbeing and sleep support.
Target patient population:
The Welldoc App is intended for individuals 18 years and older:
• T1 and T2 Diabetes
• Pre-diabetes
• Hypertension
• Heart failure
• Weight and obesity management
And support for:
- Mental wellbeing
- Sleep
Clinical Overview
Welldoc has achieved 11 510(k) clearances for diabetes functionality within its digital health platform, and an IP portfolio of 42 patents for its advanced AI and first-in-class tech. With over 75 clinical publications, Welldoc has built an extensive library of clinical research focused on the value of AI-powered digital health solutions.
Intended Use:
The Welldoc® App includes Welldoc Diabetes and Welldoc Diabetes Rx, which is Software as a Medical Device (SaMD) intended to be used by healthcare providers (HCPs) and their patients – aged 18 years and older – who have type 1 or type 2 diabetes. Welldoc Diabetes and Welldoc Diabetes Rx are intended to assist patients in managing their diabetes with guidance from their providers. Welldoc Diabetes Rx requires a prescription.
Indications for use:
The Welldoc® App combines the FDA cleared medical device functionality of Welldoc Diabetes and Welldoc Diabetes Rx with other non-FDA cleared wellness and educational/self-management products. Welldoc Diabetes is indicated for use by healthcare providers (HCPs) and their patients – aged 18 years and older – who have type 1 or type 2 diabetes. Welldoc Diabetes is intended to provide secure capture, storage, and transmission of blood glucose data as well as information to aid in diabetes self-management. Welldoc Diabetes analyzes and reports blood glucose test results and supports medication adherence. In addition, Welldoc Diabetes provides coaching messages (motivational, behavioral, and educational) based on real-time blood glucose values and trends. It includes software intended for use on mobile phones or personal computers in the home or in professional healthcare settings. The software also allows for entry of other diabetes-related healthcare information and provides educational information.
Welldoc Diabetes Rx adds the following prescription use only insulin management features:
- For bolus insulin users with type 1 or type 2 diabetes, Welldoc Diabetes Rx includes an insulin dose calculator* to allow patients to use their prescribed regimen to calculate a dose of bolus insulin for a given amount of carbohydrates and/or blood glucose value.
- For basal insulin users with type 2 diabetes, Welldoc Diabetes Rx includes an Insulin Adjustment Program (IAP) which calculates appropriate long-acting basal insulin doses for titrating insulin levels based on configuration by a healthcare provider. The healthcare provider must activate the IAP and configure it with patient-specific parameters.
- For bolus insulin users with type 2 diabetes, the Welldoc Diabetes Rx IAP calculates appropriate dose adjustments of bolus insulin based on configuration of a healthcare provider. Qualified type 2 diabetes patients are those who are not achieving glycemic targets despite optimization of their basal insulin dose or their current bolus insulin regimen.
- For premixed insulin users with type 2 diabetes, the Welldoc Diabetes Rx IAP calculates appropriate dose adjustments of premixed insulin based on the configuration of a healthcare provider. Qualified type 2 diabetes patients are those who are not achieving glycemic targets and who do not take other types of insulin. Welldoc Diabetes and Welldoc Diabetes Rx are not intended to replace the care provided by a licensed healthcare professional, including prescriptions, diagnosis, or treatment.
*Insulin Dose Calculator may also be referred to as Insulin Calculator
Other Welldoc® App Non-Diabetes Functions:
Hypertension, Heart Failure, Behavioral Health, Prediabetes
The other disease state-focused products in the Welldoc App are non-FDA-cleared wellness and educational/self-management products and DO NOT interact with Welldoc Diabetes or Welldoc Diabetes Rx functions. They are intended to help people with hypertension, heart failure, or mild mental health conditions better manage and live well. Visit www.welldoc.com for full labeling information.
Contraindications
The Welldoc App is not indicated for people with gestational diabetes or who use an insulin pump. For more information, call Customer Care at 1-888-327-5345.
Outcomes:
Welldoc’s research consists of 50+ peer-reviewed, clinical publications, studies and posters—including 3 multi-site, randomized controlled studies. Within this extensive research, Welldoc has studied the impact of BlueStar on health measures such as HbA1c, medication adherence, and better blood glucose control.
Place in therapy:
Complementary to current therapies including pharmacologic-related, diet-related, exercise-related, or knowledge-related therapy pathways.
Product Access
Product description:
The Welldoc Platform includes:
- The Welldoc App provides cardiometabolic condition digital coaching across pre-diabetes, diabetes, hypertension, heart failure, weight and obesity management, with integrated mental wellbeing and sleep support. The app connects and analyzes an individual’s vital health data to provide personalized, AI-driven digital coaching and insights.
- The Welldoc Care Management Portal provides accessible health data and actionable insights to support clinical decisions, interventions, and population health management between office visits.
- Health Reports that consolidate and analyze Welldoc App user-generated data with integrating standards of care to provide insights, trends, and patterns.
Prescription status:
Welldoc Diabetes is available over the counter (OTC) and does not require a prescription. However, Welldoc Diabetes Rx requires a prescription for additional insulin management support.
User access:
Individuals can download the Welldoc App via the App Store or Google Play Store. A program ID is provided by their health plan, health system, or employer to register for the app.
Use of this product requires access to:
- Internet or Wi-Fi for initial download and periodic syncing
- A mobile phone, tablet, or computer
Provider access:
The Welldoc App creates Health Reports that can be shared by the patient and/or the Welldoc Care Management Portal, enabling secure access to an individual’s data and insights to support care team workflows and population health management.
Coverage options:
The Welldoc Platform can be paid for by health plans, health systems, or employers.
Product availability:
The Welldoc App is available in:
- USA: FDA-cleared Class II Medical Device
- Canada: Health Canada-licensed Class II Medical Device
- Japan, UK & Australia: (coming soon)
- EU: Class IIa CE mark (MDR) (coming soon to market)
Engaging users. Driving outcomes.
Welldoc’s digital first approach uses AI-driven algorithms to provide immediate, tailored and relevant coaching to individuals when they’re making decisions about their health. The Welldoc App empowers individuals with personalized, evidenced-based feedback to help them successfully self-manage their health condition while enhancing connections to their healthcare team.
Clinical Trials
The provided set of evidence represents a sample of conducted studies. For more research, please visit https://www.welldoc.com/clinical-research/
- Study Title: WellDoc™ Mobile Diabetes Management Randomized Controlled Trial: Change in Clinical and Behavioral Outcomes and Patient and Physician Satisfaction
Study Design: Randomized controlled study; IRB-certified study
Outcomes: The clinical evidence shows a 2.0 point mean A1C reduction for individuals living with type 2 diabetes who used BlueStar. - Study Title: Glycemic Control: Impact on Physician Prescribing Behavior
Study Design: Randomized controlled study; IRB-certified study
Outcomes: The clinical evidence shows a 1.9-point mean A1C reduction for individuals living with type 2 diabetes who used BlueStar.
1 Quinn, C.C. Shardell, M.D. Terrin, M.L. Barr, E.A. Ballew, S.H. Gruber-Baldini, A.L. Cluster-randomized trial of a mobile phone personalized behavioral intervention for blood glucose control. Diabetes Care. 2011 Sep;34(9):1934-42. Erratum in: Diabetes Care. 2013 Nov;36(11):3850.
2 Kumbara A, Iyer A, Leone K, Shomali M. Evaluating the Impact of a Combined Real-Time CGM/Digital Health Solution on Glucose Control for People with Type 2 Diabetes. Poster presented at: ATTD Conference Advanced Technologies & Treatments for Diabetes, Barcelona & Online, 2022.
3 Kumbara A, Iyer A, Shomali M. Blood pressure improvement in people using a digital health solution for comprehensive diabetes self-management. Poster presented at: 16th Annual Cardiometabolic Health Congress; October 14-17, 2021; National Harbor, MD.
4 Welldoc Data on File. Subset of multiple Welldoc programs (n = 106 who paired with weight scales/data). 68% of 106 experienced an average of 6% weight loss over a duration of 6 months. Results may vary based on Welldoc App adherence.
5 IBM Watson Health. Estimating the economic value of a digital therapeutic in type 2 diabetes. Welldoc-sponsored report 2018. Published August 2018.
6 Welldoc Data on File. Results may vary based on Welldoc App adherence.