Product Highlight
Condition targeted:
Opioid use disorder (OUD)

How it works:
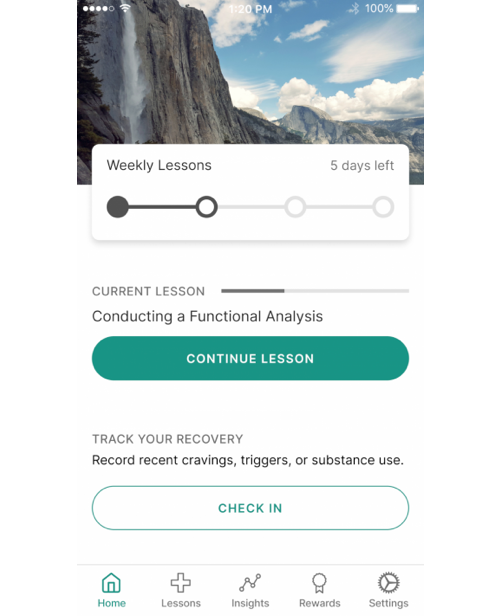
The reSET-O prescription digital therapeutic is a 12-week (84-day) software application to be used as an adjunct to outpatient treatment using transmucosal buprenorphine and contingency management to increase retention of patients with OUD. It is limited to persons with a valid prescription from their licensed provider.
Impact:
Adding reSET-O® to outpatient treatment using buprenorphine increased retention of patients with OUD by almost 15%.
Product Overview
Medical condition:
Opioid Use Disorder (OUD)
Target patient population:
Patients with OUD, as an adjunct to outpatient treatment that includes transmucosal buprenorphine and contingency management, for patients 18 years or older who are currently under the supervision of a clinician.
What to expect:
Significant improvements in retention in treatment. OUD can be managed through cognitive-behavioral approaches and contingency management-based therapies, in conjunction with MAT.
Note: for more information, please visit https://peartherapeutics.com/products/reset-reset-o/.
Clinical Overview
Indications for use:
reSET-O is intended to increase retention of patients with Opioid Use Disorder (OUD) in outpatient treatment by providing cognitive behavioral therapy, as an adjunct to outpatient treatment that includes transmucosal buprenorphine and contingency management.
Outcomes:
The treatment retention rate during the 12-week intervention was 82.4% in the reSET-O + Treatment-as Usual (TAU) group, compared to 68.4% in the TAU group alone. This difference was significant, with a p-value of 0.0224.
Directions:
reSET-O is a prescription digital therapeutic designed and engineered to deliver behavioral therapy modeled on the Community Reinforcement Approach (CRA). CRA is a specific form of cognitive behavioral therapy (CBT) designed for patients with OUD. reSET-O combines CRA (for therapy) and fluency training (to reinforce concept mastery) and should be implemented in conjunction with Contingency Management and buprenorphine treatment to help retain patients with OUD in outpatient treatment.
Risks & warnings:
reSET-O is intended for patients whose primary language is English and who have access to an Android/iOS tablet or smartphone. reSET-O is intended only for patients who own a smartphone and are familiar with use of smartphone apps (applications).
Clinicians should not use reSET-O to communicate with their patients about emergency medical issues. Patients should be clearly instructed not to use reSET-O to communicate to their clinician any urgent or emergent information. The long-term benefit of treatment with reSET-O on abstinence has not been evaluated in studies lasting beyond 12-weeks in the Opioid Use Disorder (OUD) population. The ability of reSET-O to prevent potential relapse after treatment discontinuation has not been studied.
Place in therapy:
Complementary to current outpatient treatment with buprenorphine pharmacotherapy.
Product Access
Product description:
The reSET-O prescription digital therapeutic is a 12-week (84-day) software application to be used as an adjunct to outpatient treatment using transmucosal buprenorphine and contingency management to increase retention of patients with Opioid Use Disorder (OUD). It is limited to persons with a valid prescription from their licensed provider.
Prescription status:
Yes, a prescription is required.
Patient access:
Patients download the reSET-O app through the Apple App Store or Google Play Store on their smartphone or tablet. An access code is given to them at the time of prescription.
The reSET-O app is supported on the following mobile operating systems:
- iOS
- Android
Patients should have access to a smartphone or tablet. Patients should be able to upload data periodically, i.e. have internet/wireless connection access.
Provider access:
The reSET-O Clinician Dashboard allows clinicians and other health care providers to view information about patients’ use of the reSET-O App, including lessons completed, patient-reported substance use, patient-reported cravings and triggers, patient-reported medication use, compliance rewards, and in clinic data inputs such as urine drug screen results. The reSET-O Clinician Dashboard can be used to track in-clinic data such as urine drug screen results and appointment attendance.
The reSET-O Clinician dashboard is optimized for desktop computer access and is supported on the following web browsers:
- Google Chrome
- Firefox
- Microsoft Internet Explorer
- Microsoft Edge
- Safari
Coverage options:
reSET-O is typically paid for by fully insured health plans and self-insured employers as either a pharmacy benefit or a medical benefit. Co-payments, co-insurance, and deductibles may apply.
Product availability:
reSET-O is available in:
USA: 510(k), FDA Cleared, Software as a Medical Device
Unique Features
reSET-O is the first FDA-cleared prescription digital therapeutic for OUD.
Clinical Trials
The provided set of evidence represents a sample of conducted studies. For a comprehensive collection contact manufacturers directly.
Study Title: Adding an Internet-delivered Treatment to an Efficacious Treatment Package for Opioid Dependence
Study Design: Block-randomized, unblinded, parallel, 12-week treatment trial.
Outcomes: Compared to those receiving CM-alone, CRA+ recipients exhibited, on average, 9.7 total days more of abstinence (95% confidence interval [CI = 2.3, 17.2]) and had a reduced hazard of dropping out of treatment (hazard ratio = 0.47; 95% CI [0.26, 0.85]).